What's Magnetic Moment?
What Is Magnetic Moent?
As we all know, matter is composed of atoms, and atoms are composed of atomic nuclei and electrons. In atoms, electrons have an orbital magnetic moment due to their movement around the nucleus. Electrons have spin-magnetic moments due to their spins. The magnetic moments of atoms mainly come from the electronic magnetic moments, which are the source of all material magnetism.
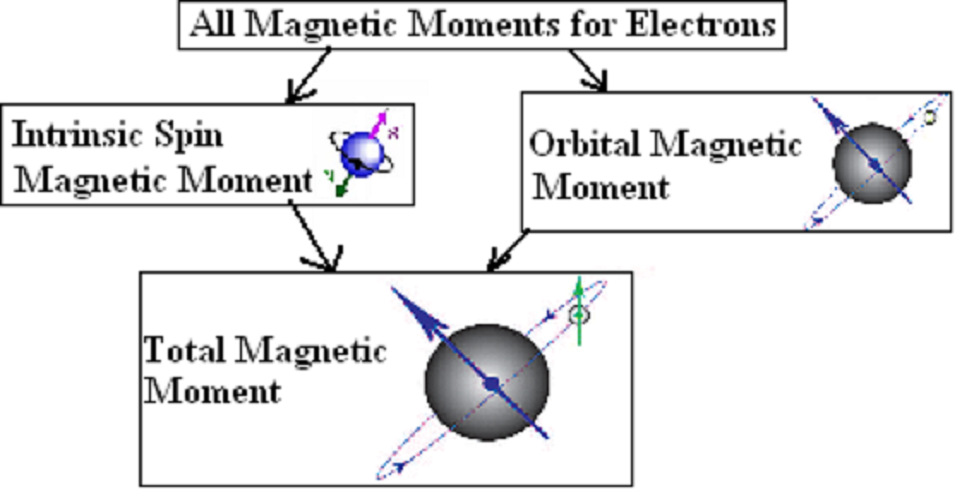
Magnetic Moment
Magnetic Moment of a Single Isolated Atom
The magnetic moment is a directional vector. There are two kinds of spin modes of electrons in atoms. In most matter, there are as many electrons with upward and downward spins, and the magnetic moments they produce will cancel each other out, and the entire atom is not magnetic to the outside. Only the atoms of a few matters have different numbers of electrons in different spin directions.
In this way, after the magnetic moments of electrons with opposite spins cancel each other out, the spin magnetic moments of some of the electrons are not canceled, so the atom has a magnetic moment. The magnetic moment of a single atom depends on the atomic structure, and the atoms of all elements in the periodic table have their magnetic moments.
Magnetic Moment of Atoms in Crystal
What we discussed above is the magnetic moment of a single atom, but in a solid crystal or amorphous, the atoms are located at the crystal node, and these atoms will be affected by the nuclear electric field and electronic electrostatic field of adjacent atoms, so the magnetic moment of an atom in a crystal is different from that of a single isolated atom.
For example, iron, cobalt, and nickel are called 3d transition metals. In the crystal, the electrons of some atoms will become common electrons of neighboring atoms. The electronic structure of the atoms will change, and part of the orbital magnetic moment will be frozen. At this time, the only contribution to the magnetic moment of the atoms in the crystal is the spin magnetic moment, so the magnetic moment of the atoms in the crystal is different from the theoretical value.
Magnetic Moment of Macroscopic Matter
Due to the different magnetic moments of different atoms, the interaction between the atomic magnetic moments of macroscopic substances is caused. The arrangement of the atomic magnetic moments at room temperature is different. We divide them into paramagnetic substances, diamagnetic substances, ferromagnetic materials, ferrimagnetic materials, and antiferromagnetic materials, these characteristics include the following three.
1. Magnetization M
The macroscopic magnetism of a substance is contributed by the magnetic moment of its atoms or molecules. We call the total magnetic moment of the material per unit volume V as the magnetization of the material, which is represented by M and the unit is A/m. Suppose the volume of a substance is V, it has n atoms, and the magnetic moment of each atom is μJ, then M=μJ1+μJ2+……+μJn, that is, M=∑μJ/V.
2. Magnetization Curve of Magnetization Intensity (M~H Curve)
When the external magnetic field is zero, the atomic magnetic moments may be arranged chaotically, but when we apply a non-zero external magnetic field, under the action of the external magnetic field, each atomic magnetic moment can be turned in the direction of the external magnetic field. At this time, the magnetization M of the substance changes. The relationship curve that the magnetization M changes with the change of the external magnetic field H is called the magnetization curve, abbreviated as the M~H magnetization curve. The magnetization curves of different substances are also different.
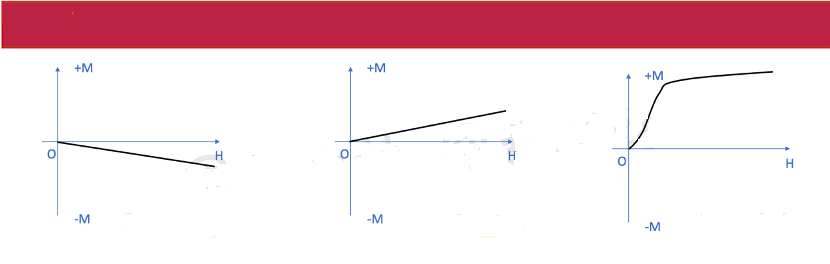
Magnetization Curves of Different Substances
3. Magnetic Susceptibility χ
On the M~H magnetization curve, the ratio of M to H at any point is called the susceptibility, which is represented by χ. Among them, the unit of M is A/m, and the unit of H is also A/m, so it is the relative magnetic susceptibility and has no unit. We use the size and arrangement of the above-mentioned atomic magnetic moments, the shape of the M~H magnetization curve, and the magnetic susceptibility parameters to describe the magnetic properties of materials and classify the materials.
Different Magnetic Materials
--Paramagnetic Substances
Paramagnetic substances are a kind of magnets that can be magnetized according to the direction of the magnetic field when they are moved closer to the magnetic field. If the external magnetic field is removed, the internal magnetic field will also return to zero, making it non-magnetic. Common paramagnetic substances such as aluminum and oxygen. Each atom of a paramagnetic substance has a magnetic moment, which makes the paramagnetic substance have an inherent atomic magnetic moment. In addition, there is no interaction between adjacent atoms of paramagnetic substances, so at room temperature, the atomic magnetic moments are arranged chaotically, and the projection value of the atomic magnetic moment μJ in any direction is zero.
When there is an external magnetic field H, the atomic magnetic moment of this type of substance can only rotate a very small angle along the direction of the external magnetic field, and its magnetization increases slowly with the increase of the external magnetic field. Its magnetic susceptibility χ>0 and the value of χ is generally 10-5~10-3. To make the atomic magnetic moments of paramagnetic substances completely aligned with the direction of the external magnetic field, according to a rough estimate, this requires an external magnetic field strength of 109-1010A/m, which is currently difficult to achieve with artificial magnetic fields.
--Diamagnetic Substance
Diamagnetic materials are materials with negative magnetic susceptibility, which means that the direction of the magnetic field after magnetization is opposite to the direction of the external magnetic field. All organic compounds are diamagnetic. Graphite, lead, water, etc. are all diamagnetic materials. The projection of the atomic orbital magnetic moment and the spin magnetic moment of the diamagnetic substance in the magnetic field is zero, which means that the diamagnetic substance has no net atomic magnetic moment.
But, under the action of the external magnetic field, the electron orbit will produce an induced additional magnetic moment, and this induced magnetic moment is opposite to the direction of the external magnetic field, so negative magnetism appears. The direction of magnetization of a diamagnetic material is negative, opposite to the external magnetic field, and its absolute value increases linearly with the increase of the external magnetic field.
--Ferromagnetic Substance
A ferromagnetic substance is a kind of magnetic substance that can maintain its magnetized state even if the external magnetic field disappears after being magnetized under the action of an external magnetic field. Iron, cobalt, and nickel are all ferromagnetic substances.
--Ferrimagnetic Substance
The macroscopic magnetism of ferrimagnetic substances is the same as that of ferromagnetic substances, except that the magnetic susceptibility is lower. A typical ferrimagnetic substance is a ferrite. The most significant difference between ferrimagnetic substances and ferromagnetic substances is the difference in the internal magnetic structure.
--Antiferromagnetic Substance
Anti-ferromagnetic materials do not generate magnetic fields, which are relatively uncommon. Most anti-ferromagnetic materials only exist in low-temperature conditions. If the temperature exceeds a certain value, it will usually become paramagnetic. For example, chromium, manganese, etc. have antiferromagnetic properties.
Conclusion
Thank you for reading our article and we hope it can help you to have a better understanding of the magnetic moment. If you want to know more about magnets, we would like to advise you to visit Stanford Magnets for more information.
Stanford Magnets is a leading magnet supplier across the world, that has been involved in R&D, manufacturing, and sales of magnets since the 1990s. It provides customers with high-quality permanent magnets like neodymium magnets, SmCo magnets, AlNiCo magnets, and ferrite magnets (ceramic magnets) at a very competitive price.















